Related Articles
- 12 Feb 20
Endometriosis is one of the most common chronic gynecological conditions in women of reproductive age. Not only is it associated with severe, debilitating pain, but it can also have significant implications for a woman’s fertility.[1]
- 28 Jan 21Advanced glycation end-products (AGEs) are compounds that are generated under hyperglycemic conditions. They could be formed endogenously, or they could be consumed from our diet. Increased presence of AGEs in the human body is associated with diverse age-related and chronic conditions...
- 12 Feb 20
Metabolic syndrome is a cluster of risk factors that increase one’s chances of developing serious illnesses in the future. Metabolic syndrome doubles the chance of developing cardiovascular disease while increasing the risk of diabetes, fatty liver, and several types of cancers.[1][2]
- 10 Jun 20
Depression and anxiety deplete your energy, desire, and hope. They make it difficult for you to take a step that can help you to feel better. Depression is the second most frequent medical condition seen in common medical practice around the world; it is caused by alterations of the neurotransmitters in our central nervous system.[1] It is a mood disorder that occurs differently in different individuals.
- 12 Feb 20
Mild traumatic brain injuries (mTBIs) and postconcussion syndrome are becoming more of a public health concern. Although termed “mild,” the effects can certainly be debilitating and severe in some cases. The symptoms of postconcussion syndrome include headaches, memory and concentration difficulties, irritability, fatigue, nausea, visual disturbances, light and noise sensitivity, judgement problems, depression, and anxiety.
- 10 Jun 20
Peripheral neuropathy is a common type of neuralgia, with diabetic neuropathy and postherpetic neuralgia being the most common types.
- 09 Jan 20
There are various supports for headache management such as supplements, acupuncture/body work, and herbal medicine.
Headaches can be divided into different categories, determining treatment type. - 08 Jul 20
Polycystic ovarian syndrome (PCOS) is one of the most common endocrine disorders. PCOS affects one in every five women of the reproductive age.
- 26 Feb 21
The kidneys are one of the body’s most important filtration systems and are sometimes overlooked until there is an issue. In traditional Chinese medicine, the kidneys are often referred to as the “root of life,” and they play an important role in overall vitality, especially as we age.
- 05 Jun 17
- 26 Feb 21
Long before February was declared “Heart Month” in Canada, “American Heart Month” in the United States, and “National Heart Month” in the United Kingdom, children and adults celebrated February 14 as a day of love and affection.
- 11 Aug 15
 There is a new, growing interest in the field of male infertility, given the recent trends of declining conception over the past decade. The times are telling of families having fewer children, later in age and often having more difficulties with conception. Infertility can pose physical, psychological, financial and economic burden on individuals, the health care system and society.
There is a new, growing interest in the field of male infertility, given the recent trends of declining conception over the past decade. The times are telling of families having fewer children, later in age and often having more difficulties with conception. Infertility can pose physical, psychological, financial and economic burden on individuals, the health care system and society. - 02 Nov 16
- 22 Dec 15
 According to Statistics Canada, results from the 2009 to 2011 Canadian Health Measures Survey (CHMS) indicate that 1 in 5 Canadian adults aged 18 to 79 had metabolic syndrome. Metabolic Syndrome, also known as Syndrome X, Insulin Resistance Syndrome, or Mets refers to a cluster of conditions that occur together. These conditions include high blood pressure, high blood sugar levels, excess body fat around the waist or mid-central obesity, and abnormal cholesterol levels.
According to Statistics Canada, results from the 2009 to 2011 Canadian Health Measures Survey (CHMS) indicate that 1 in 5 Canadian adults aged 18 to 79 had metabolic syndrome. Metabolic Syndrome, also known as Syndrome X, Insulin Resistance Syndrome, or Mets refers to a cluster of conditions that occur together. These conditions include high blood pressure, high blood sugar levels, excess body fat around the waist or mid-central obesity, and abnormal cholesterol levels. - 09 Nov 15
 Congestive heart failure (CHF) is the process that occurs when the heart does not pump blood effectively. Congestion then follows in the lungs and peripheral tissues. CHF can occur in both ventricles or just on one side, although it will often progress to both sides. The symptoms include breathlessness and difficulty breathing, usually made worse by lying down
Congestive heart failure (CHF) is the process that occurs when the heart does not pump blood effectively. Congestion then follows in the lungs and peripheral tissues. CHF can occur in both ventricles or just on one side, although it will often progress to both sides. The symptoms include breathlessness and difficulty breathing, usually made worse by lying down - 16 Jan 16
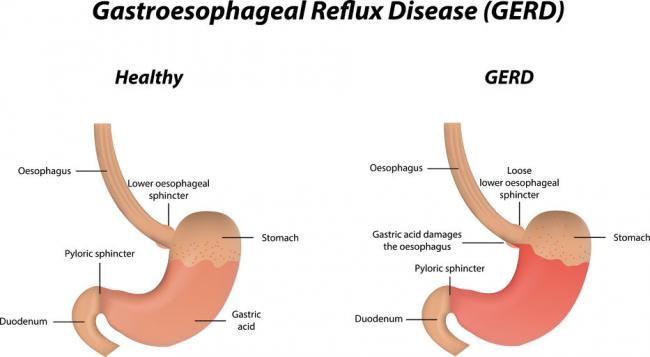
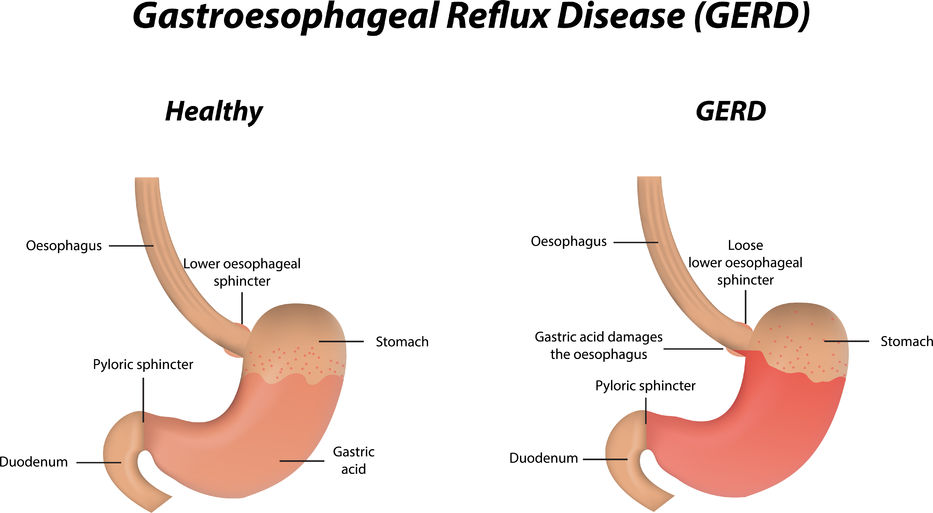 Gastroesophageal Reflux Disease or GERD can be defined as a condition of mucosal damage produced by the abnormal reflux of gastric contents into the esophagus. According to the Canadian Digestive Health Foundation, 5 million Canadians experience heartburn and/or acid regurgitation at least once each week.
Gastroesophageal Reflux Disease or GERD can be defined as a condition of mucosal damage produced by the abnormal reflux of gastric contents into the esophagus. According to the Canadian Digestive Health Foundation, 5 million Canadians experience heartburn and/or acid regurgitation at least once each week. - 13 Feb 16
 Noticing blood while brushing or flossing can be alarming and shouldn’t be ignored! The importance of oral health is a concept introduced to most Canadians at a very young age, and with good reason. The Ontario Dental Hygienist’s Association reports that the link between oral infections and other diseases in the body is becoming well-documented and accepted within the health-care community. Periodontal disease is one of the most common human diseases
Noticing blood while brushing or flossing can be alarming and shouldn’t be ignored! The importance of oral health is a concept introduced to most Canadians at a very young age, and with good reason. The Ontario Dental Hygienist’s Association reports that the link between oral infections and other diseases in the body is becoming well-documented and accepted within the health-care community. Periodontal disease is one of the most common human diseases - 22 Dec 15
 Our eyes and our sense of vision are extremely important to quality of life. As we age, our eyes tend to deteriorate, not only with regards to acuity, but also with regards to overall health. There are two conditions which are quite common and both cause problems with vision. These conditions are presented below.
Our eyes and our sense of vision are extremely important to quality of life. As we age, our eyes tend to deteriorate, not only with regards to acuity, but also with regards to overall health. There are two conditions which are quite common and both cause problems with vision. These conditions are presented below. - 13 Apr 15
 Pre-eclampsia is a condition that has life-threatening consequences if not treated immediately. If you are a pregnant woman experiencing symptoms of pre-eclampsia, it is important to seek medical attention. Although the cause remains unknown, advancements in research are being made, and several theories exist for the cause of the condition.
Pre-eclampsia is a condition that has life-threatening consequences if not treated immediately. If you are a pregnant woman experiencing symptoms of pre-eclampsia, it is important to seek medical attention. Although the cause remains unknown, advancements in research are being made, and several theories exist for the cause of the condition. - 10 Apr 16
 What Is It and What Causes It? Seasonal affective disorder (SAD) is a form of depression that occurs during certain times of the year. It generally occurs during the colder months, and is associated with shorter days. Reduced sunlight exposure during the fall and winter can disrupt the body’s internal clock, also known as the circadian rhythm.
What Is It and What Causes It? Seasonal affective disorder (SAD) is a form of depression that occurs during certain times of the year. It generally occurs during the colder months, and is associated with shorter days. Reduced sunlight exposure during the fall and winter can disrupt the body’s internal clock, also known as the circadian rhythm.
Newsletter
Most Popular
- 03 Apr 14
- 17 Jun 13
- 17 Jun 13
- 17 Jun 13
- 01 Jul 13
- 17 Jun 13
- 17 Jun 13
- 17 Jun 13
- 01 Jul 13
- 17 Jun 13
- 17 Jun 13
- 17 Jun 13
- 01 Jul 13